Abstract
This article provides a comprehensive review of current understanding regarding the etiology, risk factors, diagnosis, prevention strategies, and treatment protocols for peri-implantitis. Evidence-based approaches for both non-surgical and surgical management are critically evaluated, and a detailed case study illustrates practical implementation of contemporary treatment strategies. The importance of personalized maintenance protocols and early intervention is emphasized as essential components for long-term implant success. Understanding the multifactorial nature of peri-implantitis and implementing targeted prevention and treatment approaches is crucial for clinicians to ensure optimal outcomes in implant therapy.
Introduction
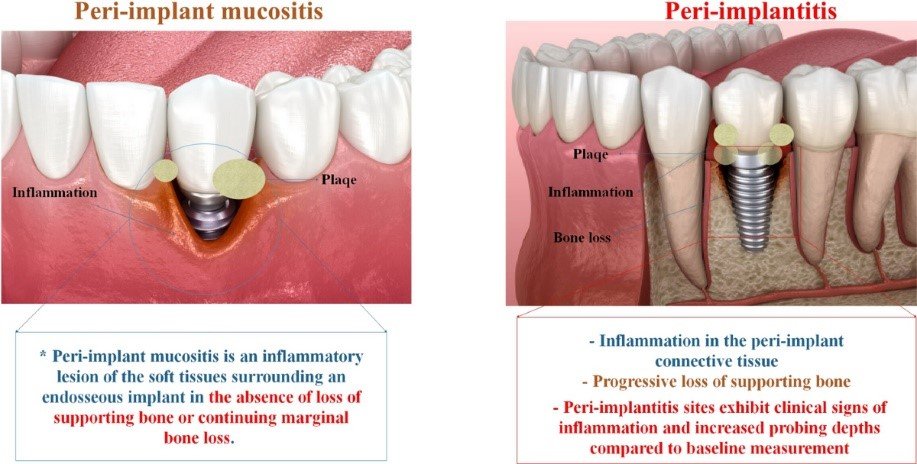
Dental implants have revolutionized the rehabilitation of edentulous patients, providing functional and aesthetic solutions with high long-term success rates. However, the increasing number of implants placed worldwide has been accompanied by a rise in biological complications, with peri-implantitis emerging as a significant challenge for clinicians (Derks & Tomasi, 2015). Peri-implantitis is characterized by inflammation in the peri-implant mucosa and progressive loss of supporting bone beyond biological bone remodeling (Schwarz et al., 2018).
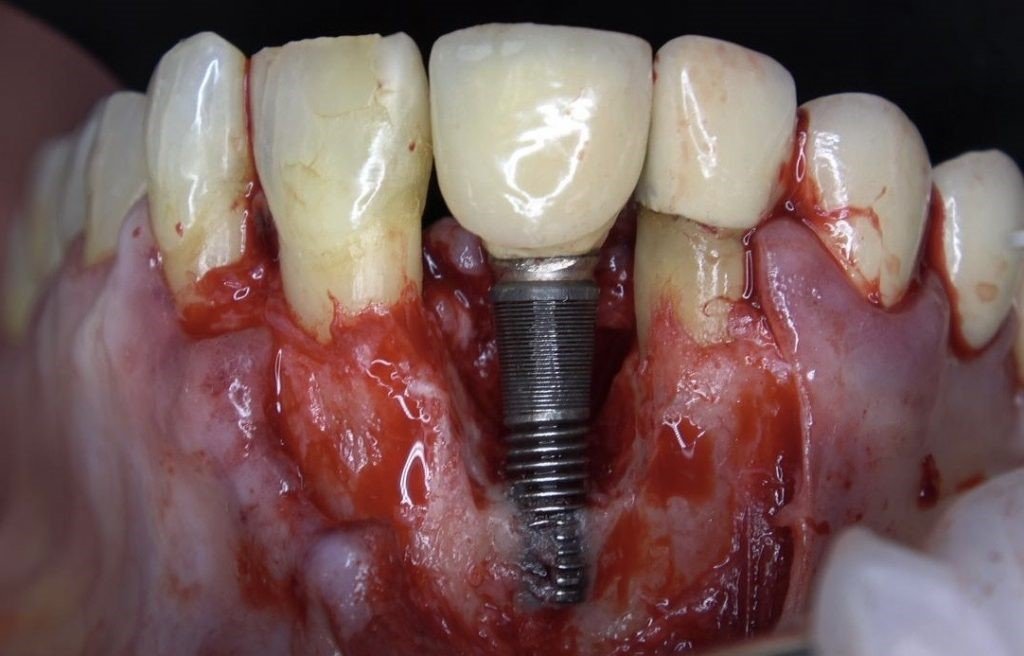
Unlike periodontitis, which typically progresses slowly, peri-implantitis often exhibits a non-linear, accelerated pattern of tissue destruction that can rapidly compromise implant stability if left untreated (Berglundh et al., 2018). The reported prevalence of peri-implantitis varies widely in the literature due to differences in case definitions and diagnostic criteria, but recent systematic reviews estimate that approximately 22% of implant patients develop peri-implantitis within 5-10 years of implant placement (Derks & Tomasi, 2015; Rakic et al., 2018).
The etiology of peri-implantitis is multifactorial, involving bacterial challenge, host response, and various risk factors. While similarities exist between periodontitis and peri-implantitis, important differences in anatomical, histological, and immunological aspects necessitate specific approaches to prevention and treatment (Carcuac & Berglundh, 2014). Understanding these distinctions is crucial for developing effective management strategies.
This article aims to provide a comprehensive review of current knowledge regarding peri-implantitis prevention and management, integrating evidence-based approaches with practical clinical considerations. A case study will illustrate the application of these principles in a challenging clinical scenario.
Etiology and Pathogenesis
Microbial Factors
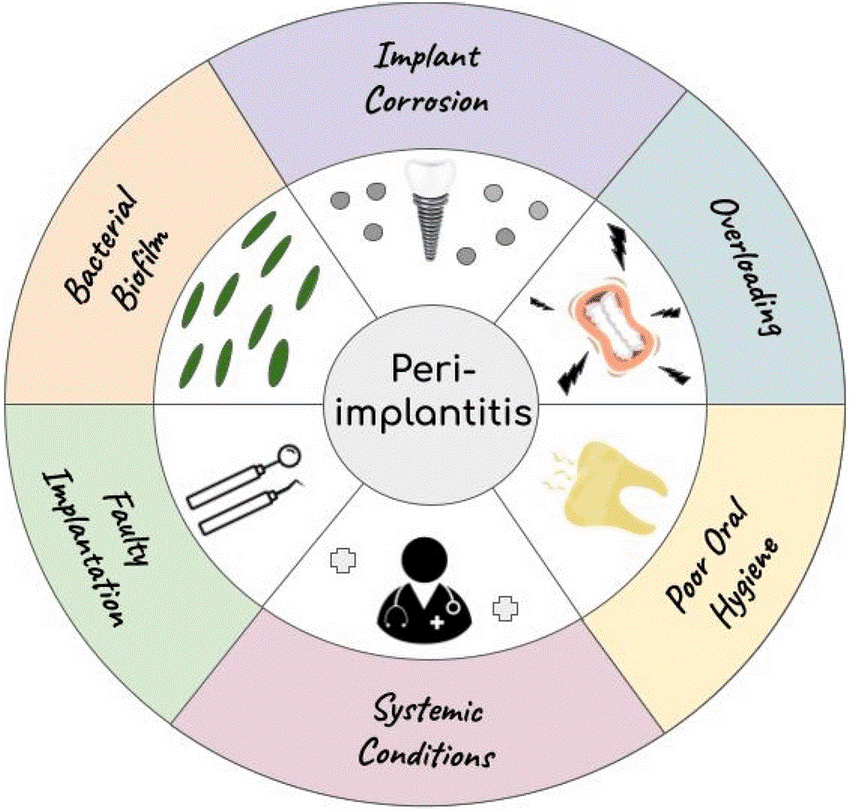
The initiation of peri-implantitis is primarily associated with bacterial biofilm accumulation on implant surfaces. Similar to periodontitis, the microbiome in peri-implantitis is characterized by a shift from predominantly Gram-positive aerobic bacteria to more pathogenic Gram-negative anaerobic species (Belibasakis, 2014). However, peri-implant microbiota exhibits greater diversity and complexity, with potentially more virulent pathogens than those found in periodontitis (Lafaurie et al., 2017).
Common pathogens associated with peri-implantitis include:
- Porphyromonas gingivalis
- Tannerella forsythia
- Treponema denticola
- Aggregatibacter actinomycetemcomitans
- Prevotella intermedia
- Fusobacterium nucleatum
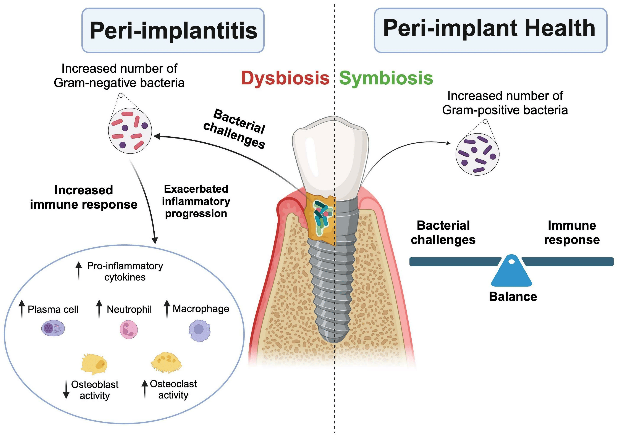
Additionally, opportunistic pathogens not typically associated with periodontitis, such as Staphylococcus aureus, Pseudomonas aeruginosa, and Candida albicans, have been isolated from peri-implantitis lesions (Rakic et al., 2016). These microorganisms can adhere to titanium surfaces and form biofilms that are resistant to host defense mechanisms and antimicrobial agents.
Host Response and Immunopathogenesis
The host inflammatory response to bacterial challenge plays a crucial role in the pathogenesis of peri-implantitis. Compared to periodontal tissues, peri-implant tissues exhibit important structural differences that influence the progression of disease:
- Absence of periodontal ligament and direct bone-to-implant contact (osseointegration)
- Parallel (rather than perpendicular) orientation of collagen fibers in the connective tissue
- Reduced vascularization in peri-implant mucosa
- Weaker epithelial attachment to the implant surface
These anatomical differences result in reduced defensive capacity against bacterial invasion, allowing more rapid apical spread of inflammation (Berglundh et al., 2011). Furthermore, the inflammatory response in peri-implantitis is characterized by a higher proportion of B-cells and plasma cells, with elevated levels of pro-inflammatory cytokines (IL-1β, TNF-α, IL-6, IL-17) and matrix metalloproteinases (MMPs) compared to periodontitis (Carcuac & Berglundh, 2014).
The implant surface itself influences the inflammatory response. Rougher surfaces, while beneficial for osseointegration, provide greater surface area for bacterial colonization and can complicate decontamination efforts once infection is established (Renvert et al., 2018).
Risk Factors
Understanding risk factors for peri-implantitis is essential for patient selection, treatment planning, and implementing preventive strategies. These factors can be categorized as patient-related, implant-related, and iatrogenic.
Patient-Related Risk Factors
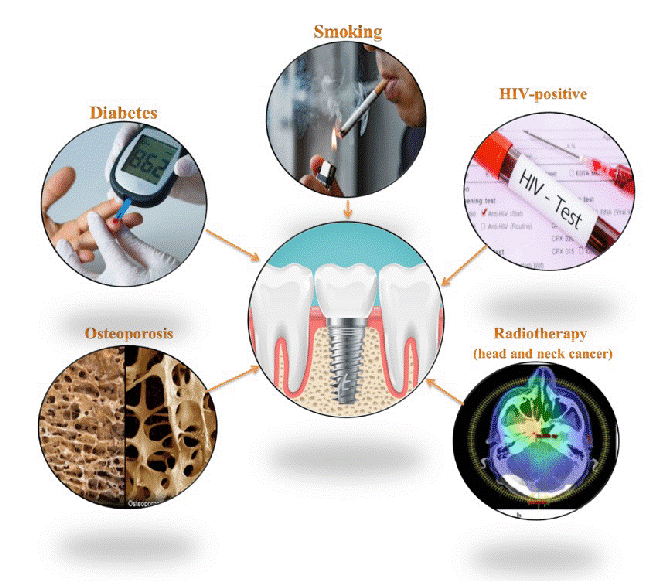
- History of Periodontitis: Strong evidence supports previous or active periodontitis as a significant risk factor for peri-implantitis, with up to 4-fold increased risk compared to periodontally healthy patients (Sgolastra et al., 2015). The microbial transfer from residual dentition to implants and a potentially hyperinflammatory host response contribute to this association.
- Poor Oral Hygiene: Inadequate plaque control is strongly associated with peri-implant diseases. Patients with plaque index >20% demonstrate significantly higher rates of peri-implantitis (Serino & Ström, 2009).
- Smoking: Tobacco use impairs wound healing, reduces vascularization, and alters immune response. Current evidence indicates that smokers have approximately 2.5 times higher risk of developing peri-implantitis than non-smokers (Stacchi et al., 2016).
- Diabetes Mellitus: Poorly controlled diabetes (HbA1c >8%) is associated with impaired healing, microvascular complications, and altered immune function, increasing susceptibility to peri-implantitis (Monje et al., 2017).
- Genetic Factors: Polymorphisms in genes regulating inflammatory mediators (IL-1, IL-6, TNF-α) may predispose certain individuals to exaggerated inflammatory responses to bacterial challenge (Laine et al., 2013).
- Systemic Medications: Medications affecting bone metabolism (bisphosphonates, antiresorptive drugs) or immunosuppressants may influence peri-implant tissue health (Guobis et al., 2016).
Implant-Related Risk Factors
- Implant Surface Characteristics: While moderately rough surfaces improve osseointegration, they also facilitate biofilm formation if exposed to the oral environment. Some studies suggest higher peri-implantitis rates with rougher surfaces (Teughels et al., 2006).
- Implant Design: Certain design features such as external hex connections, platform-matched abutments, and overcontoured restorations may contribute to marginal bone loss and subsequent peri-implantitis (Sasada & Cochran, 2017).
- Implant Position: Malpositioned implants creating emergence profiles that impede oral hygiene or establishing inadequate soft tissue thickness can predispose to peri-implantitis (Fu et al., 2017).
Iatrogenic Factors
- Cement Remnants: Excess subgingival cement from cement-retained restorations is strongly associated with peri-implantitis, acting as a nidus for bacterial colonization (Wilson, 2009). Studies have reported cement remnants in 81% of implants with peri-implantitis signs.
- Occlusal Overload: While controversial, excessive biomechanical forces may contribute to peri-implant bone loss, particularly in patients with parafunctional habits or improper prosthetic design (Chang et al., 2013).
- Inadequate Keratinized Mucosa: Limited evidence suggests that insufficient keratinized tissue (<2mm) around implants may increase susceptibility to plaque-induced inflammation and recession (Lin et al., 2013).
- Technical Complications: Loose or fractured components creating microgaps can harbor bacteria and contribute to peri-implant inflammation (Canullo et al., 2015).
Diagnosis and Classification
Early and accurate diagnosis of peri-implant diseases is crucial for successful management. The 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions established clear definitions and diagnostic criteria (Berglundh et al., 2018):
Peri-Implant Health
Characterized by:
- Absence of visual signs of inflammation
- Absence of bleeding and/or suppuration on gentle probing
- No increase in probing depth compared to previous examinations
- Absence of progressive bone loss beyond initial remodeling
Peri-Implant Mucositis
Characterized by:
- Visual signs of inflammation (redness, swelling)
- Bleeding on gentle probing (BoP)
- Possible increase in probing depth
- Absence of bone loss beyond initial remodeling
Peri-Implantitis
Characterized by:
- Visual signs of inflammation
- Bleeding and/or suppuration on gentle probing
- Increased probing depths compared to previous examinations
- Progressive bone loss beyond initial remodeling
For newly placed implants without previous examination data, a threshold of ≥6mm probing depth along with BoP and/or suppuration and bone levels ≥3mm apical to the most coronal portion of the intraosseous part of the implant is suggested for peri-implantitis diagnosis.
Diagnostic Methods
- Clinical Examination: Gentle probing (0.25N force) using plastic or metal probes to assess:
- Probing depths (comparing with baseline values)
- Bleeding on probing (BoP)
- Suppuration
- Mucosal recession
- Mobility (indicating complete loss of osseointegration in advanced cases)
- Radiographic Assessment: Standardized periapical radiographs to evaluate bone levels compared to baseline (immediately after prosthetic loading). Cone-beam computed tomography (CBCT) may provide additional information in complex cases but is not routinely recommended due to radiation exposure concerns.
- Additional Diagnostic Tools: While not essential for diagnosis, these may provide supplementary information:
- Microbial testing
- Biochemical markers in peri-implant crevicular fluid
- Genetic testing
- Resonance frequency analysis to assess implant stability
Prevention Strategies
Prevention of peri-implantitis begins before implant placement and continues throughout the patient’s lifetime. A comprehensive approach incorporating pre-implant assessment, optimal treatment planning, meticulous surgical technique, and personalized maintenance protocols is essential.
Pre-Implant Phase
- Risk Assessment and Modification:
- Complete periodontal examination and treatment of existing periodontitis
- Smoking cessation counseling
- Glycemic control in diabetic patients
- Evaluation and management of parafunctional habits
- Patient Education:
- Explanation of maintenance requirements and responsibilities
- Setting realistic expectations regarding long-term commitment
- Informed consent regarding risks and complications
- Treatment Planning Considerations:
- Strategic implant positioning to facilitate hygiene
- Consideration of bone availability and soft tissue biotype
- Selection of appropriate implant system and prosthetic design
- Decision between screw-retained versus cement-retained restorations
Surgical Phase
- Implant Selection:
- Consideration of surface characteristics appropriate for site-specific bone quality
- Platform-switching designs to preserve crestal bone
- Internal connection systems with improved bacterial sealing properties
- Surgical Principles:
- Minimally traumatic approach
- Adequate primary stability
- Appropriate positioning (3D)
- Consideration of soft tissue augmentation when keratinized tissue is inadequate
- Prosthetic Considerations:
- Screw-retained restorations when possible to eliminate cement-related risks
- If cement-retained, ensure margin placement no deeper than 1mm subgingivally
- Proper emergence profile design facilitating hygiene
- Occlusal scheme allowing axial loading and minimizing lateral forces
Maintenance Phase
- Personalized Recall Schedule:
- Typically 3-6 month intervals based on risk profile
- More frequent initially and for high-risk patients
- Professional Maintenance Protocol:
- Detailed assessment of implant and surrounding tissues
- Professional biofilm removal using appropriate instruments (carbon fiber/plastic curettes, air polishing with glycine powder)
- Reinforcement of home care techniques
- Home Care Recommendations:
- Mechanical plaque control with soft toothbrushes, interdental brushes appropriate for implant dimensions
- Supplementary chemical plaque control (0.12% chlorhexidine, essential oil rinses)
- Additional aids such as water flossers for posterior implants
Management of Peri-Implant Diseases
The management of peri-implant diseases follows a stepwise approach, with treatment strategies determined by the severity and extent of the condition. Early intervention is crucial to prevent progression and improve outcomes.
Management of Peri-Implant Mucositis
Peri-implant mucositis represents a reversible inflammatory condition and is considered the precursor to peri-implantitis. Treatment focuses on eliminating the bacterial biofilm and resolving soft tissue inflammation:
- Mechanical Debridement:
- Professional removal of biofilm using titanium-coated or carbon fiber instruments to avoid damaging implant surfaces
- Submucosal air polishing with glycine powder has shown superior efficacy compared to mechanical debridement alone (Schwarz et al., 2015)
- Chemical Adjuncts:
- Chlorhexidine irrigation (0.12-0.2%) and/or gel application
- Essential oil mouth rinses as alternatives for long-term use
- Local application of antiseptics (povidone-iodine)
- Patient Education:
- Reinforcement of oral hygiene techniques
- Demonstration of implant-specific cleaning methods
- Re-evaluation:
- Assessment after 2-4 weeks to confirm resolution
- Implementation of appropriate maintenance protocol
The success rate for treating peri-implant mucositis is approximately 90% when adequate biofilm control is achieved (Jepsen et al., 2015). Failure to resolve mucositis may indicate underlying contributing factors that require identification and management.
Management of Peri-Implantitis
The management of peri-implantitis is more complex and challenging due to implant surface characteristics that facilitate bacterial adhesion and complicate decontamination efforts. Treatment approaches can be categorized as non-surgical and surgical, with selection based on disease severity, implant characteristics, and patient factors.
Non-Surgical Therapy
Non-surgical therapy is typically the initial approach for early to moderate peri-implantitis (bone loss <2mm, probing depths <6mm):
- Mechanical Debridement:
- Similar to mucositis treatment but with emphasis on reaching the implant threads
- Use of specialized instruments designed for implant surfaces (titanium brushes, plastic curettes)
- Adjunctive Therapies:
- Local or systemic antibiotics (controversial due to resistance concerns)
- Photodynamic therapy with photosensitizers activated by specific wavelength light
- Local delivery of antimicrobials (minocycline microspheres, doxycycline gel)
- Glycine powder air polishing for biofilm disruption
- Lasers (Er:YAG) for surface decontamination
- Evaluation and Maintenance:
- Re-assessment after 1-3 months
- Decision regarding need for surgical intervention if inadequate response
Evidence suggests that non-surgical therapy alone has limited efficacy in resolving peri-implantitis, with average probing depth reductions of only 0.5-1mm and minimal radiographic bone fill (Renvert et al., 2018). Non-surgical approaches are more effective for controlling inflammation than achieving re-osseointegration.
Surgical Therapy
Surgical intervention is indicated for moderate to advanced peri-implantitis cases (bone loss >2mm, probing depths >6mm) or when non-surgical therapy fails to resolve inflammation:
- Access Surgery (Open Flap Debridement):
- Elevation of mucoperiosteal flap to access implant surface
- Thorough granulation tissue removal
- Mechanical and chemical decontamination of implant surface
- Surface Decontamination Protocols:
- Mechanical: titanium brushes, ultrasonic devices with plastic tips
- Chemical: saline, chlorhexidine, citric acid, hydrogen peroxide
- Laser therapy: Er:YAG, CO2, diode lasers
- Implantoplasty: smoothing exposed implant threads using diamond burs (in supracrestal areas)
- Regenerative Approaches:
- Indicated for contained defects with adequate walls
- Materials include:
- Bone grafts (autogenous, allogeneic, xenogeneic, alloplastic)
- Barrier membranes (resorbable or non-resorbable)
- Growth factors (rhPDGF-BB, EMD)
- PRF/PRP concentrates
- Resective Approaches:
- Indicated for horizontal bone loss and non-contained defects
- Osteoplasty to reduce bony ledges
- Implantoplasty to smooth exposed implant surfaces
- Apical positioning of flap to facilitate hygiene
- Combined Approaches:
- Integration of resective and regenerative techniques based on defect morphology
- Current evidence suggests superiority of combined approaches in complex cases
Outcomes of surgical therapy vary widely depending on defect morphology, implant surface, and treatment approach. Meta-analyses report mean probing depth reductions of 2-3mm and radiographic bone fill of 1.5-2mm with regenerative approaches (Ramanauskaite et al., 2016). However, complete resolution of peri-implantitis with re-osseointegration remains unpredictable.
Treatment Plan and Implementation
Phase 1: Initial Therapy
- Control of Predisposing Factors:
- Reinforcement of diabetes management (referral to physician)
- Oral hygiene instruction with emphasis on implant-specific care
- Smoking cessation reinforcement
- Non-Surgical Therapy:
- Removal of cement remnants using plastic curettes
- Mechanical debridement with titanium brushes
- Glycine powder air polishing (EMS AIR-FLOW®)
- Local application of 0.12% chlorhexidine gel
- Prescription of 0.12% chlorhexidine rinse twice daily for 14 days
Re-evaluation after 4 weeks showed partial improvement in soft tissue inflammation but persistent 6-7mm probing depths and bleeding on probing, indicating the need for surgical intervention.
Phase 2: Surgical Therapy
- Access Flap Surgery:
- Full-thickness mucoperiosteal flap elevated
- Complete granulation tissue removal
- Thorough implant surface decontamination:
- Mechanical: titanium brushes
- Chemical: 3% hydrogen peroxide followed by saline irrigation
- Laser decontamination: Er:YAG laser (100mJ/pulse, 10Hz)
- Regenerative Procedure:
- Defect morphology assessment revealed a contained circumferential defect
- Application of enamel matrix derivative (Emdogain®)
- Grafting with particulate xenograft (Bio-Oss®) mixed with autogenous bone chips harvested from adjacent area
- Coverage with resorbable collagen membrane (Bio-Gide®)
- Tension-free primary closure with 5-0 PTFE sutures
- Post-Surgical Protocol:
- Amoxicillin 500mg TID for 7 days
- 0.12% chlorhexidine rinse twice daily for 2 weeks
- Soft diet for 2 weeks
- Suture removal after 14 days
Phase 3: Maintenance and Monitoring
- Short-term Follow-up:
- Clinical assessment at 1, 3, and 6 months
- Radiographic evaluation at 6 months showing partial bone fill (2mm gain)
- Professional biofilm management at each visit
- Long-term Maintenance:
- Enrollment in 3-month professional maintenance program
- Reinforcement of oral hygiene at each visit
- Annual radiographic assessment
Outcome
At the 12-month follow-up, the following outcomes were observed:
- Complete resolution of inflammation with no bleeding on probing
- Reduction in probing depths to 3-4mm
- Radiographic evidence of 2.5mm bone fill
- Stable soft tissue margins without recession
- Patient reported no symptoms and demonstrated improved oral hygiene compliance
This case illustrates the importance of identifying contributing factors (cement remnants), implementing a staged approach to treatment, and establishing an effective maintenance protocol. The successful outcome was attributable to thorough decontamination of the implant surface, regenerative therapy appropriate for the defect morphology, and the patient’s improved compliance with home care and professional maintenance.
Current Trends and Future Directions
Innovative Decontamination Approaches
Recent developments in implant surface decontamination include:
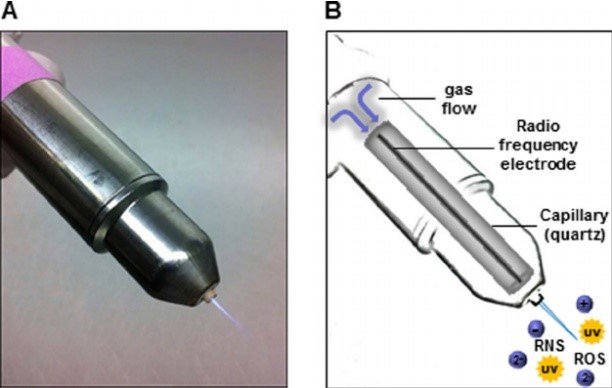
- Cold Atmospheric Plasma (CAP): Generates reactive oxygen species and nitrogen species that effectively eliminate biofilms without damaging implant surfaces (Rupf et al., 2015).
- Photodynamic Therapy (PDT): Combination of photosensitizer (typically phenothiazine chloride) and low-level laser therapy to generate reactive oxygen species that disrupt bacterial cell membranes (Sivaramakrishnan & Sridharan, 2018).
- Electrochemical Decontamination: Application of low-intensity electric current to titanium implants, creating an antimicrobial environment through electrochemical reactions (Mohn et al., 2011).
Advanced Regenerative Approaches
Emerging regenerative strategies include:
- Growth Factor Combinations: Synergistic effects of multiple growth factors (rhPDGF-BB, rhBMP-2, rhFGF-2) to enhance bone regeneration and soft tissue healing (Urban et al., 2019).
- Cell-Based Therapies: Autologous mesenchymal stem cells or bone marrow aspirate concentrate to enhance regenerative outcomes (Alvarez et al., 2018).
- 3D-Printed Custom Scaffolds: Patient-specific scaffolds designed to precisely fit peri-implant defects, potentially improving regenerative outcomes (Asa’ad et al., 2016).
Implant Surface Innovations
Development of novel implant surfaces to reduce peri-implantitis risk:
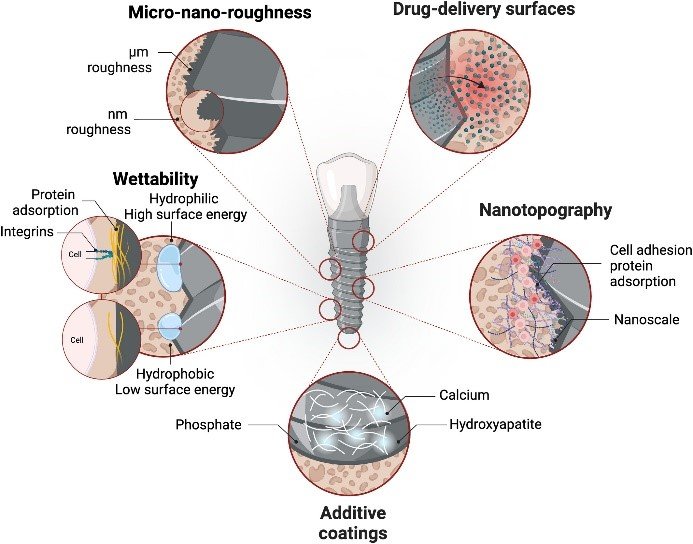
- Antimicrobial Surfaces: Incorporation of antimicrobial agents (silver, copper) or antibiotics into implant surfaces to prevent bacterial colonization (Gosau et al., 2016).
- Bioactive Surfaces: Surfaces that release bioactive molecules promoting osseointegration while inhibiting bacterial adhesion (Granato et al., 2019).
- Self-Cleaning Surfaces: Hydrophilic surfaces that resist protein adsorption and bacterial attachment (Gittens et al., 2014).
Diagnostic Advancements
Emerging diagnostic technologies include:
- Biomarker Analysis: Point-of-care devices for analyzing inflammatory mediators in peri-implant crevicular fluid to detect early disease (Alassy et al., 2019).
- Optical Coherence Tomography: Non-invasive imaging to assess peri-implant soft tissues and early bone changes (Ikeda-Artacho et al., 2019).
- Digital Technologies: Integration of digital workflows, including intraoral scanning and computer-assisted diagnosis, to improve monitoring and early detection (Rechmann et al., 2019).
Conclusion
Peri-implantitis represents a significant challenge in implant dentistry, with potential to compromise long-term implant survival and patient satisfaction. Current evidence supports a multifactorial etiology involving microbial, host-related, and iatrogenic factors that must be considered in developing preventive and therapeutic strategies.
Treatment should follow a structured approach based on disease severity, with non-surgical therapy typically preceding surgical intervention in a stepped care model. Surface decontamination represents a critical component of all therapeutic approaches, though complete re-osseointegration remains an elusive goal with current techniques.
As implant dentistry continues to evolve, ongoing research into innovative preventive strategies, improved diagnostic methods, and advanced therapeutic approaches will further enhance our ability to effectively manage peri-implant diseases. Implementation of evidence-based protocols and personalized risk-based maintenance programs remains essential for optimizing long-term implant outcomes and patient satisfaction.
References
- Alassy, H., Parachuru, P., & Wolff, L. (2019). Peri-Implantitis Diagnosis and Prognosis Using Biomarkers in Peri-Implant Crevicular Fluid: A Narrative Review. Diagnostics, 9(4), 214.
- Alvarez, P., Hee, C. K., Solchaga, L., Snel, L., Kestler, H. K., Lynch, S. E., & Hollinger, J. O. (2018). Growth factors and craniofacial surgery. Journal of Craniofacial Surgery, 29(1), 46-51.
- Asa’ad, F., Giannì, A. B., Giannobile, W. V., & Rasperini, G. (2016). 3D-printed scaffolds and biomaterials: Review of alveolar bone augmentation and periodontal regeneration applications. International Journal of Dentistry, 2016, 1239842.
- Belibasakis, G. N. (2014). Microbiological and immuno-pathological aspects of peri-implant diseases. Archives of Oral Biology, 59(1), 66-72.
- Berglundh, T., Armitage, G., Araujo, M. G., Avila-Ortiz, G., Blanco, J., Camargo, P. M., Chen, S., Cochran, D., Derks, J., Figuero, E., Hämmerle, C. H. F., Heitz-Mayfield, L. J. A., Huynh-Ba, G., Iacono, V., Koo, K. T., Lambert, F., McCauley, L., Quirynen, M., Renvert, S., … Zitzmann, N. (2018). Peri-implant diseases and conditions: Consensus report of workgroup 4 of the 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions. Journal of Clinical Periodontology, 45(S20), S286-S291.
- Berglundh, T., Zitzmann, N. U., & Donati, M. (2011). Are peri-implantitis lesions different from periodontitis lesions? Journal of Clinical Periodontology, 38(s11), 188-202.
- Canullo, L., Penarrocha-Oltra, D., Covani, U., Botticelli, D., Serino, G., & Penarrocha, M. (2015). Clinical and microbiological findings in patients with peri-implantitis: A cross-sectional study. Clinical Oral Implants Research, 27(3), 376-382.
- Carcuac, O., & Berglundh, T. (2014). Composition of human peri-implantitis and periodontitis lesions. Journal of Dental Research, 93(11), 1083-1088.
- Chang, M., Chronopoulos, V., & Mattheos, N. (2013). Impact of excessive occlusal load on successfully-osseointegrated dental implants: A literature review. Journal of Investigative and Clinical Dentistry, 4(3), 142-150.
- Derks, J., & Tomasi, C. (2015). Peri-implant health and disease: A systematic review of current epidemiology. Journal of Clinical Periodontology, 42(S16), S158-S171.
- Fu, J. H., Hsu, Y. T., & Wang, H. L. (2017). Identifying occlusal overload and how to deal with it to avoid marginal bone loss around implants. European Journal of Oral Implantology, 10(Suppl 1), 91-103.
- Gittens, R. A., Scheideler, L., Rupp, F., Hyzy, S. L., Geis-Gerstorfer, J., Schwartz, Z., & Boyan, B. D. (2014). A review on the wettability of dental implant surfaces II: Biological and clinical aspects. Acta Biomaterialia, 10(7), 2907-2918.
- Gosau, M., Haupt, M., Thude, S., Strowitzki, M., Schminke, B., & Buergers, R. (2016). Antimicrobial effect of different intracanal medications on various bacterial species. European Journal of Dentistry, 10(1), 69-73.
- Granato, R., Marin, C., Suzuki, M., Gil, J. N., Janal, M. N., & Coelho, P. G. (2019). Biomechanical and histomorphometric evaluation of a thin ion beam bioceramic deposition on plateau root form implants: An experimental study in dogs. Journal of Biomedical Materials Research Part B: Applied Biomaterials, 107(1), 73-79.
- Guobis, Z., Pacauskiene, I., & Astramskaite, I. (2016). General diseases influence on peri-implantitis development: a systematic review. Journal of Oral & Maxillofacial Research, 7(3), e5.
- Ikeda-Artacho, M. C., Figuero, E., Briz-Sebastián, M., Noguerol, F., Lázaro, A., Sicilia, A., & Herrera, D. (2019). Non-invasive optical technologies for diagnosis
